Wasu Chaitree
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Boorata Dechpisutthithum
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Winrath Khrutchan
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Muthita Kongroiyou
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Chiwathan Srimangkorn
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Ratthammanoon Sakulsinghdusit
Department of Chemical Engineering, Faculty of Engineering and Industrial Technology, Silpakorn University, Nakhon Pathom 73000, Thailand
Joongjai Panpranot
Center of Excellence on Catalysis and Catalytic Reaction Engineering, Department of Chemical Engineering, Faculty of Engineering, Chulalongkorn University, Bangkok 10330, Thailand
Keywords: Bismuth, Cobalt, Electro-oxidation of glycerol, Nickel
Abstract
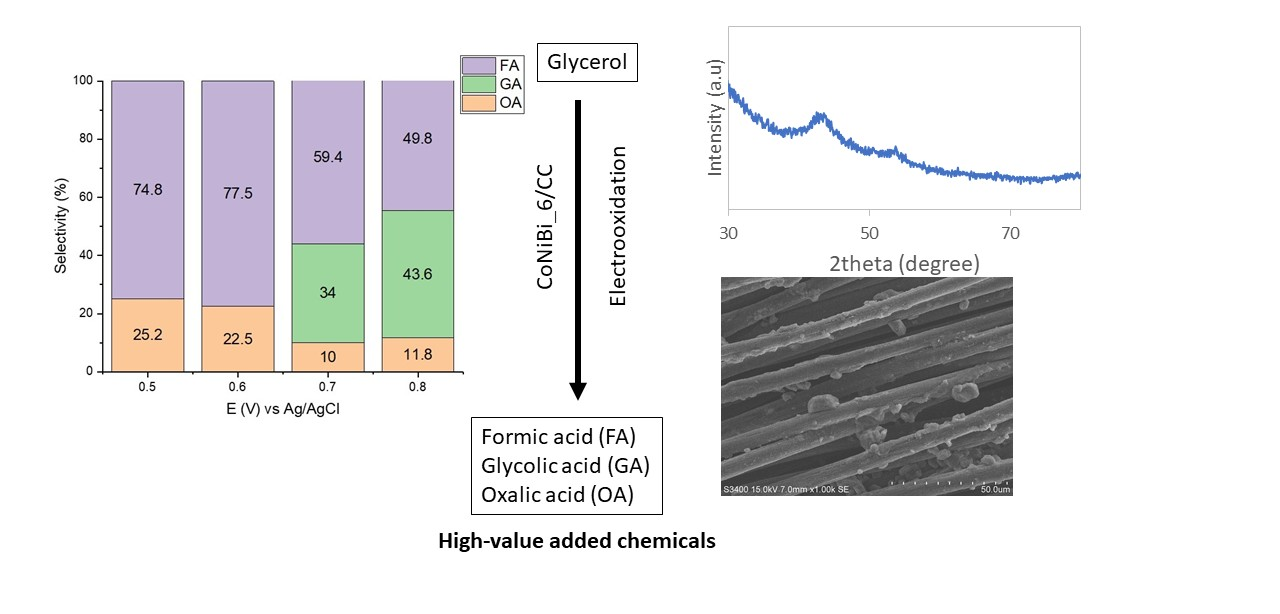
Toward the development of non-noble metal electrocatalysts for the valorization of glycerol, the effect of the addition of Bi on Co-Ni electrocatalysts supported on carbon cloth (CC) for the electro-oxidation of glycerol (EOG) was investigated. The CoNiBi/CC electrocatalysts were prepared via an electroless deposition method and characterized using X-ray diffraction, scanning electron microscopy, energy-dispersive X-ray spectroscopy, and X-ray photoelectron spectroscopy. Their electrochemical activity was investigated using cyclic voltammetry, linear sweep voltammetry, and chronoamperometry. The effect of the Bi (NO3)3 concentration in the electroless bath on the EOG performance was studied, finding that CoNiBi/CC prepared using 6 mM Bi (NO3)3 (CoNiBi_6/CC) provided higher current density and lower onset potential than other electrocatalysts. The effect of applied potentials on the formation of products was also investigated. The analysis of the liquid products using high-performance liquid chromatography showed that formic acid (FA) was obtained as the main product along with oxalic acid as a minor product. The highest rate of FA formation was 772.95 mol cm−2 h−1 at 0.7 V vs. Ag/AgCl after 2 h of reaction, and the highest selectivity for FA was 77.5% at 0.6 V vs. Ag/AgCl. Overall, the CoNiBi/CC electrocatalyst is a promising and cost-effective alternative for the electrochemical valorization of glycerol into value-added chemicals.
References
Pagliaro MV, Bruni F, Oberhauser W, Capozzoli L, Berretti E, Bartoli F. Hydrogen and high-value-added chemicals from glycerol electroreforming using a highly efficient and selective ligand-stabilized PdCu catalyst. ACS Sustainable Chem Eng. 2025;13(13):4975–4987.
Sarkar A, Tyagi T, Sharma S, Joshi V, Gogoi P, Puzari A. Synthesis of glycerol carbonate from glycerol and CO2 over Cu/In2O3/ZnO nanostructured catalyst. Catal Today. 2025;448:115164.
Terekhina I, WhiteAnn J, Johnsson C. Electrocatalytic oxidation of glycerol to value-added compounds on Pd nanocrystals. ACS Appl Nano Mater. 2023;6(13):11211–11220.
Massaneiro J, Valério TL, Pellosi DS, Silva BJGd, Vidotti M. Electrocatalytic oxidation of glycerol performed by nickel/cobalt alloys: Adding value to a common subproduct of chemical industry. Electrochim Acta. 2024;506:145013.
Habibi B, Delnavaz N. Electrooxidation of glycerol on nickel and nickel alloy (Ni-Cu and Ni-Co) nanoparticles in alkaline media. RSC Adv. 2016;6:31797-31806.
Houache MSE, Safari KH, Botton GA, Baranova EA. Modification of nickel surfaces by bismuth: Effect on electrochemical activity and selectivity toward glycerol. ACS Appl Mater Interfaces 2020;12(13):15095–15107.
Wang K, Hong L, Liu Z-L. The role of Bi3+ complex ion as the stabilizer in electroless nickel plating process. AIChE J. 2009;55(4):1046-1055.
Domańska AJ, Skitał PM. Electrodeposition of alloy nanostructures (Co-Ni) in the presence of sodium benzene sulfonate (SBS) and their application in alkaline hydrogen evolution. Molecules. 2025;30:1771.
Yin X, Li H, Yuan R, Lu J. Hierarchical self-supporting sugar gourd-shape MOF-derived NiCo2O4 hollow nanocages@SiC nanowires for high-performance flexible hybrid supercapacitors. J Colloid Interface Sci. 2021;586:219-232.
Afzali N, Keshavarzi R, Tangestaninejad S, Gimenez S, Mirkhani V, Moghadam M. Multifunctional approach to improve water oxidation performance with MOF-based photoelectrodes. Appl Mater Today. 2021;24:2-4.
Yang X, Wang D, Yu R, Bai Y, Shu H, Ge L. Suppressed capacity/voltage fading of highcapacity lithium-rich layered materials via the design of heterogeneous distribution in the composition. J Mater Chem A. 2014;2:3899-3911.
Li Z, Bian C, Hu L. Exploration of the corrosion behavior of electroless plated Ni-P amorphous alloys via x-ray photoelectron spectroscopy. Molecules. 2023;28:377-380.
Singh S, Sahoo RK, Shinde NM, Yun JM, Mane RS, Chung W. Asymmetric faradaic assembly of Bi2O3 and MnO2 for a high-performance hybrid electrochemical energy storage device. RSC Adv. 2019;9:32154–32164.
Hu E, Yao Y, Chen Y, Cui Y, Wang Z, Qian G. Boosting hydrogen generation by anodic oxidation of iodide over Ni-Co (OH)2 nanosheet arrays. Nanoscale Adv. 2021;3:604–610.
Şahin EA, Kardaş G. Cobalt-modified nickel–zinc catalyst for electrooxidation of methanol in alkaline medium. J Solid State Electrochem. 2013;17:2871–2877.
R. Ortiz, Márquez OP, Márquez J, Gutiérrez C. Necessity of oxygenated surface species for the electrooxidation of methanol on iridium. J Phys Chem B. 1996;100(20): 8389–8396.
Radi AE, Ashour WFD, Elshafey R. Glycerol electrocatalytic oxidation on nickel hydroxide nanoparticles/poly‑eriochrome black t modified electrode. Electrocatalysis. 2022;13:653–662.
Rozario A, Silva RKSe, Freitas MBJG. Recycling of nickel from NiOOH/Ni (OH)2 electrodes of spent Ni–Cd batteries. J Power Sources. 2006;158:754–759.
Barakat NAM, Khalil KA, Mahmoud IH, Kanjwal MA, Sheikh FA, Kim HY. CoNi bimetallic nanofibers by electrospinning: Nickel-based soft magnetic material with improved magnetic properties. J Phys Chem C. 2010;114:15589-15593.
Lima VS, Almeida TS, Andrade ARD. Glycerol electro-oxidation in alkaline medium with Pt-Fe/C electrocatalysts synthesized by the polyol method: Increased selectivity and activity provided by less expensive catalysts. Nanomater. 2023;13:1173-1190.
Zalineeva A, Baranton S, Coutanceau C. How do Bi-modified palladium nanoparticles work towards glycerol electrooxidation an in situ FTIR study. Electrochim Acta. 2015;176:705-717.
Jeffery DZ, Camara GA. The formation of carbon dioxide during glycerol electrooxidation in alkaline media: First spectroscopic evidences. Electrochem commun. 2010;12(8):1129-1132.
Wang C-Y, Yu Z-Y, Li G, Song Q-T, Li G, Luo C-X. Intermetallic PtBi nanoplates with high catalytic activity towards electro-oxidation of formic acid and glycerol. Chem Electro Chem. 2020;7(1):239-245.
Houache MSE, Hughes K, Safari R, Botton GA, Baranova EA. Modification of nickel surfaces by bismuth: Effect on electrochemical activity and selectivity toward glycerol. ACS Appl Mater Interfaces 2020;12(13):15095–15107.
Coutanceau C, Baranton S, Kouamé RSB. Selective electrooxidation of glycerol into value-added chemicals: A Short Overview. Front Chem. 2019;7:1-15.
Nascimento AA, Alencar eM, Zanata CR, Teixeira-Neto E, Mangini APM, Camara GA. First assessments of the influence of oxygen reduction on the glycerol electrooxidation reaction on Pt. Electrocatalysis. 2019;10:82-94.

License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
